
Scientists who specialize in chemistry are called chemists.Ĭhemistry is used all around us. 
Sometimes chemistry is called the "central science" because it is an important part of other major sciences such as biology, Earth science, and physics.  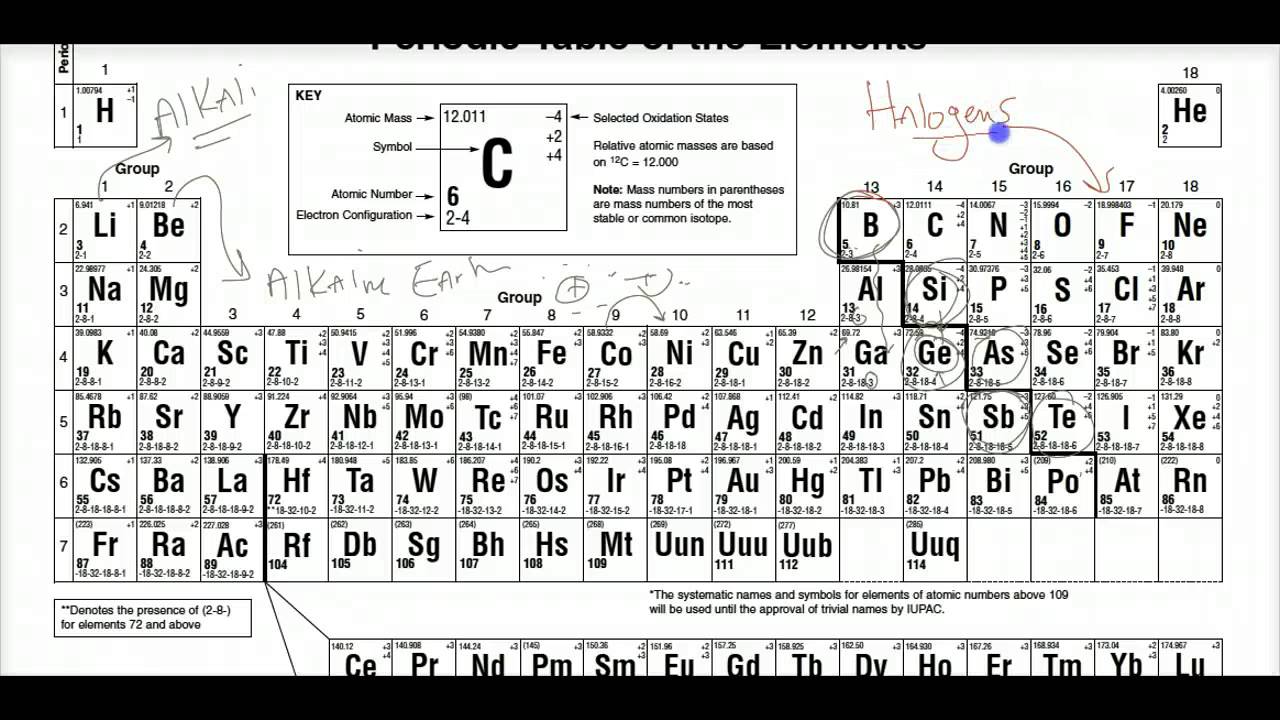
Chemistry is considered a physical science and is closely related to physics. Ligand that coordinates to one central metal through coordinate bonds from two different atoms binary acidĬompound that contains hydrogen and one other element, bonded in a way that imparts acidic properties to the compound (ability to release H+ ions when dissolved in water) binary compoundĬompound containing two different elements.Chemistry is the branch of science that studies the properties of matter and how matter interacts with energy. Salt of the hydrogen carbonate ion HCO3- bidentate ligand Single or series of galvanic cells designed for use as a source of electrical power bicarbonate anion Reaction involving the transfer of a proton from water to a base, yielding hydroxide ions and the conjugate acid of the base base ionization constant (Kb)Įquilibrium constant for a base ionization reaction basicĪ solution in which < battery Metal oxide that behaves as a base towards acids base ionization Location in a trigonal bipyramidal geometry in which there is another atom at a 180° angle and the equatorial positions are at a 90° angle base anhydride Reaction between identical species yielding ionic products for water, this reaction involves transfer of protons to yield hydronium and hydroxide ions axial position Procedure in which the electron configuration of the elements is determined by “building” them in order of atomic numbers, adding one proton to the nucleus and one electron to the proper subshell at a time autoionization Solution for which water is the solvent Aufbau principle Molecular orbital located outside of the region between two nuclei electrons in an antibonding orbital destabilize the molecule aqueous solution Species that can act as either an acid or a base anodeĮlectrode in an electrochemical cell at which oxidation occurs antibonding orbital Species that may either donate or accept a proton in a Bronsted-Lowry acid-base reaction amphoteric Solid material such as a glass that does not have a regular repeating component to its three-dimensional structure a solid but not a crystal amphiprotic Two or more forms of the same element, in the same physical state, with different chemical structures amorphous Primary battery similar to a dry cell that uses an alkaline (often potassium hydroxide) electrolyte designed to be an improved replacement for the dry cell, but with more energy storage and less electrolyte leakage than typical dry cell alkaline earth metal (also, actinoid series) actinium and the elements in the second row or the f-block, atomic numbers 89–103 active electrodeĮlectrode that participates as a reactant or product in the oxidation-reduction reaction of an electrochemical cell the mass of an active electrode changes during the oxidation-reduction reaction alkali metal Inner transition metal in the bottom of the bottom two rows of the periodic table actinide series Weak acid or base whose conjugate partner imparts a different solution color used in visual assessments of solution pH acidicĪ solution in which > actinide Reaction involving the transfer of a proton from an acid to water, yielding hydronium ions and the conjugate base of the acid acid ionization constant (Ka)Įquilibrium constant for an acid ionization reaction acid-base indicator Molecular orbital in which the electron density is found along the axis of the bond π* bonding orbitalĪntibonding molecular orbital formed by out of phase side-by-side overlap of atomic orbitals, in which the electron density is found on both sides of the internuclear axis, and there is a node between the nuclei σ* bonding orbitalĪntibonding molecular orbital formed by out-of-phase overlap of atomic orbital along the axis of the bond, generating a node between the nuclei acid anhydrideĬompound that reacts with water to form an acid or acidic solution acid ionization Molecular orbital formed by side-by-side overlap of atomic orbitals, in which the electron density is found on opposite sides of the internuclear axis σ bonding orbital
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
